BiomimeticPublished on 05.06.2024
Breakthrough in membrane technology could impact multiple industries
Researchers from the Adolphe Merkle Institute (AMI), together with international collaborators, have pioneered a novel method for creating thin, energy-converting membranes that mimic the structure and function of biological cell membranes. This discovery could have significant applications in fields ranging from implantable artificial electric organs to water desalination.
The new technique leverages the interface of an aqueous two-phase system to form and stabilize these membranes. By carefully controlling the conditions under which two immiscible water-based solutions interact with the opposing sides of these membranes, the researchers created membranes that are just 35 nanometers thick but can cover areas larger than 10 square centimeters without defects. “This approach takes advantage of favorable interactions to stabilize ultra-thin self-assembled structures that are at least one thousand-fold larger than was previously possible,” says Assistant Prof. Alessandro Ianiro, a former group leader in AMI’s Biophysics lab.
The method employs block copolymers (BCPs), highly tunable polymers consisting of two or more distinct polymer segments, to form a bilayer at the interface of the two phases. The resulting membranes exhibit remarkable mechanical properties and self-healing capabilities, making them robust and durable for practical use.
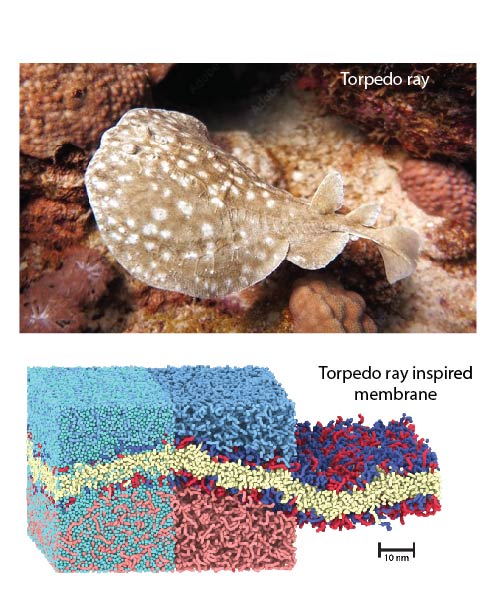
These artificial membranes replicate the selective ion transport functions of natural cell membranes. By incorporating a natural transport peptide, the membranes achieve high ion selectivity, allowing them to generate electric power from solutions of different salts. This functionality is inspired by the electric organs of rays and other electric fish, which use similar principles to generate power.
This development, reported in the leading journal Nature, could have significant applications in various fields. In energy storage, these membranes could enable the development of large-scale devices to store electrical energy. In water desalination, they may provide highly selective barriers that efficiently separate ions from water. In medical treatments such as dialysis, the membranes' ability to selectively filter ions could lead to more efficient and less invasive procedures. Finally, these membranes may enable implantable electric power sources that are recharged continuously from the body’s metabolic energy.
“This advancement takes our previous aspirations to develop fish-inspired artificial electric organs a significant step closer towards biocompatible power sources. Ultimately our goal is that these human-made systems will closely mimic – and interact with – the complex functions of biological organisms,” says AMI’s Chair of Biophysics, Prof. Michael Mayer.
Future iterations of these membranes, possibly incorporating more efficient ion channels, could achieve performance levels comparable to the electric organ of biological systems like the electric ray. The scalability of these membranes also suggests that they could be prepared with large areas to increase the transport rate across them and that they could be stacked to create larger systems for high-value separations or power generation and storage.
This research was led by AMI’s Biophysics group in collaboration with the Sustainable Functional Polymers group of Nico Bruns at the Technical University of Darmstadt, a Theory and Computer Simulation group at the University of Paris-Saclay, the Bio- and Nano-Instrumentation lab at EPFL, as well as the Polymer Chemistry and Soft Matter Physics groups at AMI.
References:
Sproncken et al., “Large-area, self-healing block copolymer membranes for energy conversion” Nature, 2024. DOI: 10.1038/s41586-024-07481-2. https://www.nature.com/articles/s41586-024-07481-2
Guha et al., “Powering electronic devices from salt gradients in AA-battery-sized stacks of hydrogel-infused paper” Advanced Materials, 2021.
Schroeder et al., “An electric-eel-inspired soft power source from stacked hydrogels” Nature, 2017.